Quantitative DNA analysis and other applications of real-time PCR
The polymerase chain reaction (PCR) revolutionized the forensic individualization of biological material by forming the basis of the so-called “genetic fingerprint”, which allows for e.g. comparisons between stains found at a crime scene and a suspect, or the identification of human remains. PCR based genotyping approaches became indispensable tools in modern forensic science, routine case-work, and paternity testing. Not surprisingly, a constant evolution of this methodology is to be witnessed.
One important example for these technological advancements is real-time PCR [Heid 1996], which gained steadily more and more importance over the last decade. From the kinetic information gained by recording the product-accumulation related fluorescent signal during the extension phase of each PCR cycle, inferences can be made on the DNA content of the sample under investigation. Endpoint fluorescence levels can also be used for the typing of single nucleotide polymorphisms.
Real-time PCR is state-of-the art for both the quantification of nucleic acids and the analysis of sequence variation in a homogeneous assay format. It is automation and high-throughput friendly, offers unprecedented sensitivity and specificity, shows a very large linear dynamic range, and its homogeneous assay format makes error-prone, time and labor intensive post-PCR manipulations unnecessary. Target-specific fluorogenic detection chemistries (e.g. TaqMan probes) and the multicolor capability of recent real-time PCR instruments enable the simultaneous quantification of different targets, saving valuable sample, time, and labor.
Accurate knowledge of the amount of amplifiable template DNA in unknown samples can greatly increase the chances for success in PCR-based genotyping. In forensic investigations, evidence material collected at crime scenes frequently contains PCR-inhibiting substances and/or suffered from environmental stress, leading to a sometimes severe decrease in the number of amplifiable template DNA molecules for downstream PCR-based genotyping. Non-PCR-based quantification methods are often cumbersome, not human specific, and not suitable to address PCR inhibition or DNA degradation. With real-time quantitative PCR (rtqPCR), however, the amount of DNA is determined by the same principal technique (i.e. PCR) that also forms the basis of almost all current genotyping approaches. Hence, the obtained results are more likely to predict the effects of unintentionally co-extracted inhibitors, and to reflect the actual amount of amplifiable template molecules present in unknown samples, than those obtained with non-PCR-based methods. Information regarding PCR inhibition and the amount of amplifiable target molecules with a minimum length required for a particular downstream application helps not only to increase the success rate for DNA typing, but might also prove useful for the assessment of the reliability of the genotyping results, especially when analyzing low copy number samples.
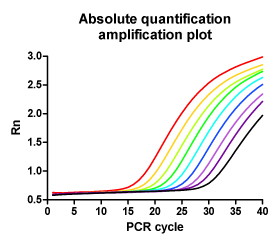
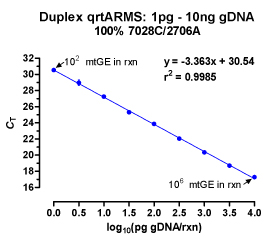
In a previous study [Niederstätter 2007] we designed on the basis of a duplex rtqPCR TaqMan assay published by [Andréasson 2002] a modular real-time PCR concept for nuclear DNA/ mitochondrial DNA quantification and PCR inhibitor detection by means of an internal amplification positive control. Modules with differing amplicon sizes for a particular target DNA were converted into each other by simply substituting one or both amplification primers. With a set of only two hybridization probes, this approach allows to easily switch between simultaneous measurement of nuclear DNA and mitochondrial DNA, determination of the degree of DNA fragmentation, and detection of PCR inhibition causing potential false negative results. This information helps to optimize the use of valuable samples, analysis time and lab-resources by aiding the selection of the most appropriate typing strategy [Berger 2009].
Development of real-time PCR quantification assays for human DNA is an ongoing task at our institute. We are particularly interested in pushing the detection and quantification limits of nuclear DNA into the lower sub-picogram range without losing the ability to asses DNA fragmentation and PCR inhibition. This can be principally achieved by aiming at multi-locus genetic elements [e.g. Walker 2005] instead of single copy genes as quantification-targets.
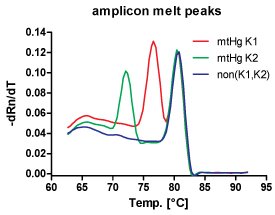
Another field of interest lies in the typing of known single nucleotide polymorphisms to study medically relevant sequence variants or as a pre-screening tool for the fast, cost efficient exclusion of samples in large-volume studies. The toolbox used contains both, allele-specific TaqMan hybridization probes [e.g. Niederstätter 2006] and allele specific amplification primers together with 2nd and 3rd generation fluorescent dyes binding specifically to double stranded DNA [e.g. Parson 2004]. Beyond mere genotyping, important aspects of these assays such as linear dynamic range, applicability for absolute DNA quantification, detection of single- or multi-contributor DNA mixtures (e.g. point-heteroplasmy, mixed stains, pooled samples) and relative quantification of the allele proportions are evaluated.
Besides working on DNA targets also relative quantification of mRNA species in a non-forensic context was performed in cooperation with other groups [e.g. Chabicovsky 2003].
Literature cited
Heid C 1996 Genome Res 6 986
Niederstätter H 2007 Forensic Sci Int Genet 1 29
Andréasson H Biotechniques 33 402
Berger C 2009 Forensic Sci Int Genet doi: 10.1016/j.fsigen.2009.01.011
Walker J 2005 Anal Biochem 337 89
Niederstätter H 2006 Int J Legal Med 120 18
Parson W 2004 Hum Mutat 24 474
Chabicovsky M 2003 Toxicol Appl Pharmacol 190 25
Collaboration partners
Michael D. Coble, Thomas J. Parsons, Armed Forces DNA Identification Laboratory (AFDIL), Rockville, MD 20850, U.S.A. present address: The International Commission on Missing Persons (ICMP), Sarajevo, Bosnia and Herzegovina.
Niels Morling, Karsten Nielsen, Department of Forensic Medicine, University of Copenhagen, Copenhagen, Denmark.